D the sharing of electrons. PO43-Which statement is true about crystal lattice energy.

Why Aldehydes Are More Reactive Than Ketones Nucleophilic Addition Reaction Electron Donating Groups Ketones Ap Chemistry
5 rows Ionic bonding is a form of chemical connection in which one atom loses valence electrons and.

. The sharing of electrons between atoms. The transfer of electrons. The atom that loses the electrons becomes a positively charged ion cation while the one that gains them.
An ionic bond is best described as A the attraction between 2 nonmetal atoms B the transfer of electrons from one atom to another. What best describes how an ionic bond forms. B the transfer of electrons from one atom to another.
C the attraction that holds the atoms together in a polyatomic ion. 1 An ionic bond is best described as A the sharing of electrons. C the attraction that holds the atoms together in a polyatomic ion.
A bond between a nonmetal and a nonmetal. They do this by hydrating the ions. A bond formed when 2 atoms share electrons.
When drawing out Lewis structures how many electrons surround the most stable. This exchange results in a more stable noble gas electronic configuration for both atoms involved. An ionic bond is best described as.
Chemical Bonds Are Best Described as Get link. A metal and nonmetal react to form an ionic bond. Ionic bonding is a type of chemical bond in which valence electrons are lost from one atom and gained by another.
B the transfer of electrons from one atom to another. C the attraction that holds the atoms together in a polyatomic ion. An ionic bond is best described as A the attraction between 2 nonmetal atoms B the transfer of electrons from one atom to another.
Which chemical species can easily form an ionic bond with a cation. C the attraction that holds the atoms together in a polyatomic ion. E the attraction between 2 metal atoms.
Ionic bonds are formed between a metal and a non-metal. Ionic bonding also commonly called electrovalent bonding is a type of chemical bonding that involves the bonding formed by the electrostatic attraction between atoms with a high electronegative difference as seen in group 1 atoms and group 7 atoms. Covalent bonds are bonds formed between two non-metals.
D the attraction between two nonmetal atoms. C the attraction that holds the atoms together in a polyatomic ion. The ionic bond is the attraction between positive and negative ions in a crystal and compounds held together by ionic bonds are called ionic compounds.
When drawing out Lewis structures how many electrons surround the most. C the attraction that holds the atoms together in a polyatomic ion. 15 Describe the relationship between electronegativity and.
Option C is correct. Other Apps - April 08 2022 Chemical Bonds Venn Diagram Chemical Bond Graphic Organizers Venn Diagram H 2 Hydrogen Gas Covalent Bond Bonds In Biology Weak Bonds Hydrogen Bonds Attraction Between And Hydrogen Bond Covalent Bonding Chemistry Basics. Which of these best describes an ionic bond.
An electrostatic attraction between oppositely charged ions. E the attraction between 2 metal atoms. An ionic bond is based on attractive electrostatic forces between two ions of opposite charge.
B the transfer of electrons from one atom to another. It is a measure of the strength of. An ionic bond involves a metal that transfers one or more electrons to a non-metal.
Covalent bonds are formed when atoms share on or more electrons. An ionic bond is best described as a the sharing of electrons. Ionic bond also called electrovalent bond type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound.
2 A covalent bond is best described as A the sharing of electrons between. C the attraction that holds the atoms together in a polyatomic ion. The bond between potassium and oxygen is best.
A the sharing of electrons. An ionic bond is best described as A the attraction between 2 nonmetal atoms B the transfer of electrons from one atom to another. An ionic bond is best described as a attraction between 2 nonmetal atoms b the transfer of electrons from one atom to another.
19 An ionic bond is best described as. E the attraction between two metal atoms. Such a bond forms when the valence outermost electrons of one atom are transferred permanently to another atom.
D the attraction between two nonmetal atoms. The C Cl bond is best described as___________. A sodium atom can only form one ionic bond because it only has one electron in its outer shell to donate.
A force that holds two oppositely charged ions together. E the attraction between 2 metal atoms. An ionic bond is best described as A the sharing of electrons.
Ionic bond definition the electrostatic bond between two ions formed through the transfer of one or more electrons. C the attraction that holds the atoms together in a polyatomic ion. A bond between gaseous elements.
Covalent polar covalent and ionic bonds Difficulty. D sharing of electrons. A bond between two metal atoms.
Ionic bonds could be best described as. B the transfer of electrons from one atom to another. 1 An ionic bond is best described as A the sharing of electrons.
D the sharing of electrons. D the sharing of electrons. This bond is the result of the nonmetal desiring electrons to fill out its valence and a metal.
A covalent bond can be best described as a bond between to atoms which. An ionic bondassociation is best described as a. D the attraction between two nonmetal atoms.
D the attraction between 2 nonmetal atoms.

Chemistry For Kids Chemical Bonding Chemistry For Kids Covalent Bonding Chemistry

The Evolutionary Process That Leads To The Formation Of A New Species From An Existing One Is Termed As Speciation Buzzle P Science Lessons Biology Stem Basic

Science Homework Comic By Eiyecaieyre On Deviantart Chemistry Classroom Science Homework Teaching Chemistry
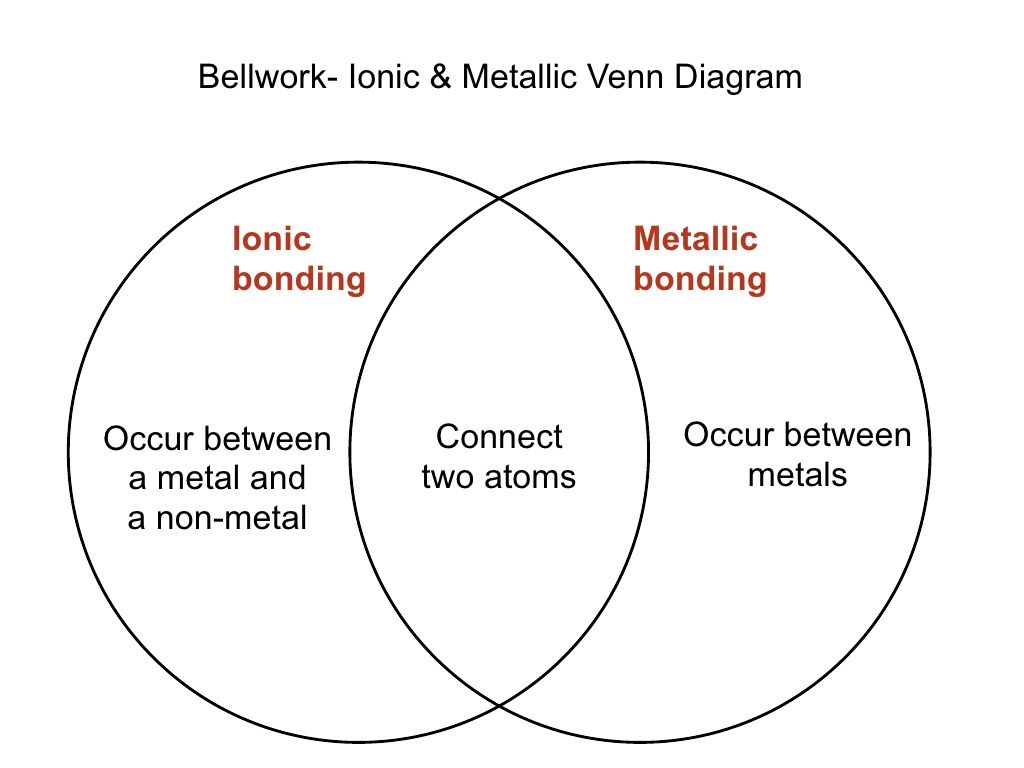
Lecture 81 Ionic Vs Covalent 2265011 By Mary Beth Smith Via Slideshare Physical Science High School Physical Science Science Chemistry
0 Comments